Questions
1. Draw and label the basic parts of a Helium atom and make sure that you show the electrical charge of those parts of the atom that are charged.
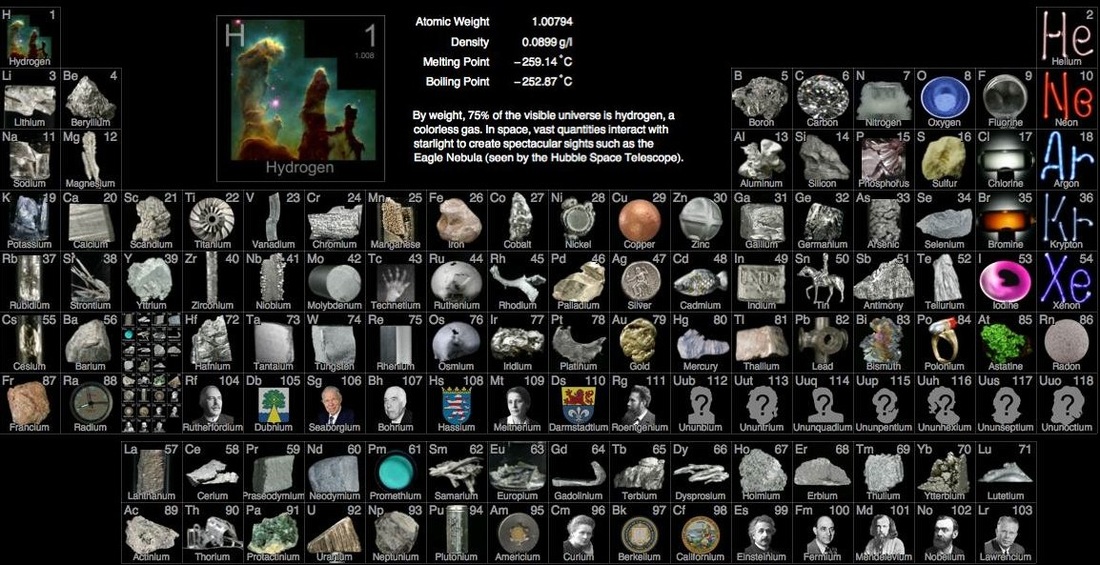
2. What is the atomic number of Iron (abbreviated Fe on the periodic table)?
3. What is the name of the element whose atomic number is 28?
4. What is the main way to tell the difference between one element and another?
5. Is there anything that you notice about the elements 26, 27 & 28? Do they remind you of anything?
2. What is the atomic number of Iron (abbreviated Fe on the periodic table)?
3. What is the name of the element whose atomic number is 28?
4. What is the main way to tell the difference between one element and another?
5. Is there anything that you notice about the elements 26, 27 & 28? Do they remind you of anything?
II. Atoms
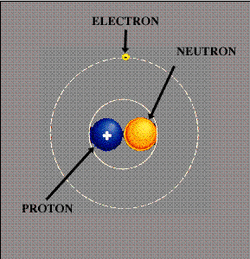
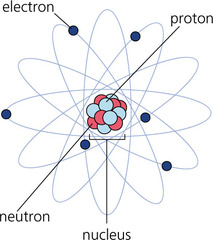 Model of atom: The electrons don't really orbit the nucleus like our planets orbit the sun. See the picture on right for a little more accurate model
A. Atoms are the basic building blocks of ordinary matter.
B. Atoms can join together to form molecules*, which in turn form most of the objects around you. C. All atoms are made of protons and electrons. Almost all atoms also contain neutrons 1. Protons are positively charged (+) and large 2. Electrons are negatively charged (-) and much smaller than protons 3. Neutrons are large and have no charge 4. Protons and neutrons are in the nucleus, while electrons surround the nucleus * Please note: that individual atoms can join together to form molecules. For example, 2 Hydrogen atoms and 1 Oxygen atom join to form H2O, or water. Two Oxygen atoms can join to for the type of oxygen you breathe, which is O2. BOTH are molecules, because 2 or more atoms are joined together. BUT, in these examples, only oxygen, whether it is made of 2 oxygens joined together in a molecule or only 1 oxygen is an element because it is made of only one type of atom - oxygen! H2O is not an element because it is made of 2 types of atoms - H and O. So, how big is an atom. The answer is that it is very, very, very, very, very small. How small? Watch the video below to find out!
|
Learning about atoms
Periodic Table This website teaches about atoms, but it becomes more advanced as it goes on. You are not required to read this, but you certainly may (and you can take a quiz afterward): http://www.chem4kids.com/files/atom_intro.html |
What is an element?

D. What is an element? 1. An element is a substance that is made entirely from one type of atom (ex. silver or gold) 2. An element is defined by the number of protons in its nucleus a. Each element has an 'atomic number', which is the number of protons in the nucleus i. Hydrogen (H) has one proton, it's atomic number is 1 ii. Ununoctium (Uuo) has 118 protons iii. Calcium has 20 protons E. Periodic Table of Elements 1. The Periodic Table of Elements shows all of the elements known to man. 2. There are 90 naturally occurring kinds of elements. Scientists have been able to make about 25 more. |
Hunting the Elements

PBS aired a special on the elements. It runs for 2 hours and is excellent to watch. We are only briefly covering atoms and elements as an introduction to the movement of electrons, however, you might enjoy watching it. Click here to go the PBS website and then click on the "Watch Program" button on the upper right.
|