Introduction
Last week we studied the basic structure of an atom and learned that an atom with more electrons than protons will have an overall negative charge. Atoms with more protons than electrons will have an overall positive charge. Atoms that have a charge, whether positive or negative are called "ions." If you are not comfortable with this topic make sure you play some of the games on "Build an Atom" before moving on to electricity.
An important thing to note: Protons do not move!! They remain in the nucleus. Only electrons move or transfer from one atom to another. And it's the movement of electrons that creates electricity.
An important thing to note: Protons do not move!! They remain in the nucleus. Only electrons move or transfer from one atom to another. And it's the movement of electrons that creates electricity.
Questions
Please read the questions on your GC assignment BEFORE reading this page. It will help to know what I'm asking for so you can look for the answers or the information that would help you figure out the problems.
Even though this is not a Google Classroom question, you should be able to fully explain why a negatively charged object is attracted to a neutral object and why, when John Travoltage is charged with electrons, the electrons jump to the metal door handle.
Even though this is not a Google Classroom question, you should be able to fully explain why a negatively charged object is attracted to a neutral object and why, when John Travoltage is charged with electrons, the electrons jump to the metal door handle.
III. What is electricity?
A. Electricity is a form of energy that happens when electrons (e-) are lost by some atoms and acquired by others
B. When e- move from a "normal" (or neutrally charged atom), the atom becomes positively charged 1. A positively charged atom attracts a negatively charged atom C. There are two main forms of electricity 1. Static electricity: the imbalance of positive & negative charges a. When you rub two objects together that are good insulators (such as a balloon with hair or wool) the wool gives its electrons to the balloon, causing the balloon to become negatively charged b. Negatively charged objects attract to the positive charges in an object, even if the object is neutrally charged (see the balloon interactive below) i. Ex. Balloons or Styrofoam rubbed on hair or wool sticks to a wall ii. Laundry sticking together after being in the dryer c. Opposite charges attract, like charges repel d. Excess electrons discharge (or burst out) in an uncontrolled way i. Ex. Lightning ii. Ex. "Zapping" your friend after going down a plastic slide |
Playing with static electricity
The Phet Interactives we've been playing with in class are being reconfigured so that most computers and IPADs can play them. Some of the older versions require that you have JAVA, so don't worry if you can't run these. Give it a try, though!
Conductors & Insulators
|
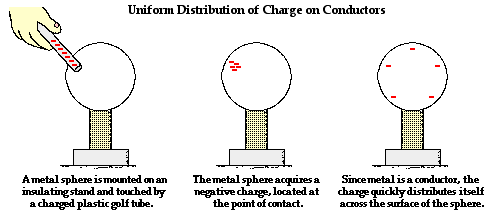
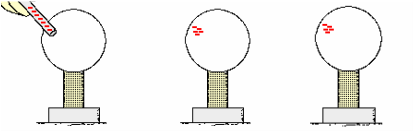
e. Materials that readily share e- from atom to atom are good conductors of electricity.
i. e- transferred to an object at a given location quickly move across entire surface of the object ii. Metal wires (especially copper)
iii. Water with minerals & salts dissolved in it iv. Humans (made up of mostly salt water) |
f. Materials that hold tightly onto their e- are not good conductors and are called insulators
i. If an e- is transferred to an insulator at a given location, the excess e- will stay at that initial location of charging ii. Ex. wool, plastic or rubber balloons, styrofoam
iii. Ex. glass iv. Ex. wood |
Don't forget to do the interactive on the left!
Remember that when we brought a good conductor such as impure water or metal in contact with the negatively-charged Styrofoam, these good conductors provided a pathway for the Styrofoam's excess electrons to move to the ground. The Styrofoam lost its electrons and therefore became neutrally-charged once again. On high humidity days (when there is a lot of water vapor in the air), the excess electrons on our Styrofoam have a pathway to move to the conductive water. That is why our Styrofoam eventually fell off the cabinet door. In past years, our Styrofoam remained on the cabinet door for weeks because the air was very low in humidity. Since both the Styrofoam and the wood are good insulators, the electrons generally stay (are 'static' or don't move from) where they were placed until a conductor provides a pathway for the electrons to "discharge." But on years when we have a lot of rain (high humidity days), the Styrofoam will fall off more quickly. This is also why you don't experience static electric shocks from going down the plastic slide as often on humid days than on dry days.
Remember that when we brought a good conductor such as impure water or metal in contact with the negatively-charged Styrofoam, these good conductors provided a pathway for the Styrofoam's excess electrons to move to the ground. The Styrofoam lost its electrons and therefore became neutrally-charged once again. On high humidity days (when there is a lot of water vapor in the air), the excess electrons on our Styrofoam have a pathway to move to the conductive water. That is why our Styrofoam eventually fell off the cabinet door. In past years, our Styrofoam remained on the cabinet door for weeks because the air was very low in humidity. Since both the Styrofoam and the wood are good insulators, the electrons generally stay (are 'static' or don't move from) where they were placed until a conductor provides a pathway for the electrons to "discharge." But on years when we have a lot of rain (high humidity days), the Styrofoam will fall off more quickly. This is also why you don't experience static electric shocks from going down the plastic slide as often on humid days than on dry days.
Triboelectric Series
|
When we rub two different materials together, which becomes positively charged and which becomes negative? Scientists have ranked materials in order of their ability to hold or give up electrons. This ranking is called the triboelectric series.
A list of some common insulator materials is shown on the right. Under ideal conditions, if two materials are rubbed together, the one higher on the list should give up electrons to the one lower on the list and become positively charged. Note: you do NOT have to memorize the list! You only have to know that materials higher on the list will give up their electrons to those lower on the list. |
TRIBOELECTRIC SERIES
your hand glass your hair nylon wool fur silk paper cotton hard rubber polyester polyvinylchloride plastic |
How Static Electricity is "Discharged"
If you play with John Travoltage's feet, he is acquiring electrons from the wool rug. Because he is a good conductor, the electrons will spread over his body. They do not have a path to the ground because he is wearing insulating rubber soles on his shoes. As he approaches the metal door knob, the excess electrons will jump to the conductive metal, which provides a pathway for the electrons. He does not actually have to touch the door knob, just be near enough for the electrons to jump or "discharge" across the gap.