Google Classroom Questions Part 1
To answer the questions below, you only need to read up to and including the notes on "recrystallization." We'll be watching several of the videos and doing the interactives in class.
|
Google Classroom Questions Part II
Later, there will be a "Part 2" that covers the material starting with "D. The shape or structure of the lattice pattern is called the crystal system."
|
I. Crystals
A. Crystals are:
1. Solid materials 2. Made up of elements, molecules or compounds (from this point on we'll call these 'particles') of a single type a. An element is made up of only one type of atom (these can be found on the Periodic Table) i. Ex. carbon or C = graphite, diamond; gold or Au; silver- Ag; sulfur – S b. Molecules are 2 or more atoms chemically connected together i. O2 is 2 oxygen atoms joined together ii. H2O is composed of two hydrogen and one oxygen atom joined together (Both H2O and O2 are molecules since they are 2 or more atoms joined together) ii. Compounds are (also) molecules, but they are composed of two or more different types elements that are chemically joined together (i.e., H2O is both a molecule AND a compound, O2 is only a molecule) (All compounds are molecules, but not all molecules are compounds) 3. Arranged in a regular, repeating 3-dimensional pattern called a lattice a. The shape of the lattice pattern is called the crystal system See the images of halite and snow crystal lattices on the right. We'll learn more about crystal systems near the bottom of this page. 4. May be microscopic or can grow in large crystals (see photo on right) B. Crystals form when an element, molecule or compound (particles) arrange symmetrically in solid form. 1. The process of crystal formation is called crystallization 2. Individual particles travel to the lattice and attach in orderly, repeating fashion, causing growth. 3. Crystals grow as long as the fluid form of the particle (whether dissolved in solution or melted) is available and there is enough space for them to grow. There is one exception that does NOT require a fluid form: recrystallization (the 3rd method of crystallization) which is describe below |
|
|
C. Crystallization occurs in one of 3 main ways:
1. Solidification: The liquid form of the particle cools and solidifies (goes from liquid to solid state) Note that this does NOT involve a solution, but it does involve a liquid! a. Ex. liquid water cools to form ice or snowflakes As you know, a liquid has to cool to become a solid. Do not confuse this with precipitation from solution by cooling. |
Liquid cooling and solidifying
|
Solutions and Saturation
Before we discuss the 2nd method of crystallization (Precipitation from Solution), you first need to learn some terms:
- Solution = a homogeneous (evenly mixed) mixture of solute and solvent (ex. salt or sugar dissolved in water)
- Solute = the thing that is dissolved into the solvent (ex. salt)
- Solvent = the thing that the solute is dissolved in (ex. water)
- Concentration = the amount of solute that is dissolved in the solvent. It is calculated by dividing the amount of solute (typically in grams) by the amount of solvent (typically in milliliters) Ex. 5 grams sugar divided by 10 ml water = 0.5 g/ml
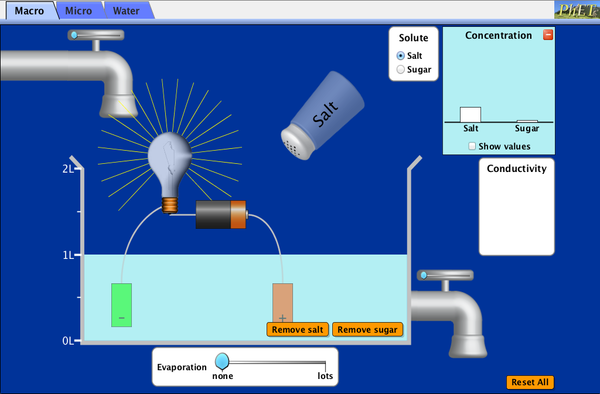
- Solubility = the most solute that can be dissolved in a solvent at any given temperature (every substance is different); Solubility is expressed as the saturation concentration (or point), or the maximum amount of solute that can be dissolved in a certain amount of solvent. A solution is "saturated" when you cannot dissolve any more solute. After it is saturated, you can add more solute, but it will not dissolve. It will simply precipitate to the bottom of the beaker. The Phet interactive below demonstrates this. We also demonstrated this when we were pouring borax into water.
- Precipitation = is when one phase (solid or liquid) is ejected from another. So, 1) a liquid ejects a solid (i.e., your salt crystals "falling" out of solution), and 2) a gas ejects a liquid (ex. rain falling from the sky) or a solid (i.e. hail or snow)
Different solutes have different saturation concentrations
In the interactive below, you will see that you can dissolve a lot more drink mix in water than some of the other solutes such as potassium permanganate. This has to do with the composition of the molecules. I do not expect you to understand why this is, but you can see this demonstrated in the interactive below.
There are 4 things you should notice when you play with this interactive:
NOTE! In this interactive, the temperature remains constant.
There are 4 things you should notice when you play with this interactive:
- There is a point where the concentration does not increase even though you are adding more solute. This is called the saturation point or saturation concentration. Once you've reached it, you can add more solute, but it won't dissolve ... it will simply precipitate to the bottom. Remember that concentration is calculated by dividing the amount of solute dissolved by the amount of solvent the solute is dissolved in. This is typically expressed in grams of solute per milliliter of solvent (g/ml), but, in the interactive below, it is expressed in moles per liter.
- The saturation point is different for each solute.
- If you remove (or evaporate) solvent, the concentration will go up until you reach saturation again.Once you do, precipitate will appear.
- If you add solvent, the concentration will go down. If it is less than the saturation point, there will be no precipitate.
NOTE! In this interactive, the temperature remains constant.
2. Precipitation from solution: When the solution a particle is dissolved in a) cools and/or b) evaporates, the crystal falls out of (or precipitates from) solution when the saturation point is exceeded.
a. Ex. halite (salt) experiment (by evaporation)
b. Ex. Borax crystal experiment (by cooling)
c. Gypsum crystals (cooling, see video link below)
NOTE: "Precipitation from solution" is the 2nd MAIN way that crystallization can occur. But, there are TWO WAYS that saturation is exceeded: either evaporation or by cooling of the solution. Note that HOW the saturation point is exceeded is different when the solution evaporates compared to when it is cooled (see explanations below)
a. Ex. halite (salt) experiment (by evaporation)
b. Ex. Borax crystal experiment (by cooling)
c. Gypsum crystals (cooling, see video link below)
NOTE: "Precipitation from solution" is the 2nd MAIN way that crystallization can occur. But, there are TWO WAYS that saturation is exceeded: either evaporation or by cooling of the solution. Note that HOW the saturation point is exceeded is different when the solution evaporates compared to when it is cooled (see explanations below)
Precipitation from Solution by EVAPORATION
a) In Precipitation from solution by evaporation, the solvent evaporates, which lowers the volume of solvent without changing the amount of solute. So, as the solvent evaporates, the concentration of solute in the remaining solvent increases. If it increases past the saturation concentration, the excess solute precipitates in the form of crystals.
Note that if the temperature remains the same, the saturation point does not change.
Note that if the temperature remains the same, the saturation point does not change.
Learn how salt & sugar dissolves in water and then recrystallizes when the water evaporates
|
Many computers no longer run this Phet interactive called Sugar & Salt Solutions. I have a copy on my computer that we'll go over in class. After viewing this together, the most important thing this helps you to visualize is:
|
Salt crystals forming by Precipitation from Solution by Evaporation
|
Salt crystal experiment: When we made halite (salt) crystals, we made a solution of halite, water, ammonia, and Mrs. Stewart's bluing. The halite was the solute and the rest was essentially* the liquid solvent.
Over several days we watched how the solvent evaporated (turned from a liquid to a gas), which caused the concentration of the solution to go up past the saturation point - so precipitation occurred. The more the solvent evaporated, the more crystals were formed. Once the solvent was entirely evaporated, all of the halite had precipitated and crystals stopped growing. This experiment demonstrated "Precipitation from solution by evaporation." *Mrs. Stewart's bluing also contains a "suspension" of blue dye molecules which will also precipitate from solution as the solvent evaporates. |
|
The time lapse example of salt crystals on the right demonstrates how crystals form as the solvent evaporates. Again, as the solvent evaporates, the concentration of the solution increases past the saturation point and the solute (salt) precipitates, thus forming crystals.
|
|
Precipitation from Solution by COOLING
b) In Precipitation from solution by cooling, precipitation of the solute into crystals happens when a warm solution cools, causing the saturation concentration to change. The warm solution may already be at saturation, but it doesn't have to be. Because the solvent is less dense at higher temperatures, there's more space in between the solvent molecules for the solute to dissolve into. So, you can dissolve more solute at higher temperatures than at lower temperatures. In other words, the saturation point is higher at higher temperatures and lower at lower temperatures. So, as a solution cools, the density of the solvent increases. This means there is less space for the solute to dissolve. Therefore, at lower temperatures, the saturation point decreases. If the solution reaches saturation, any excess solute will precipitate from solution.
NOTE that saturation in this method is achieved by changing the saturation point. The saturation point in the evaporation method does not change if the temperature remains the same.
For a more detailed example, read about our borax experiment below.
NOTE that saturation in this method is achieved by changing the saturation point. The saturation point in the evaporation method does not change if the temperature remains the same.
For a more detailed example, read about our borax experiment below.
Precipitation from Solution by Cooling - Borax experiment
Borax experiment:
In general, you can dissolve more solute at higher temperatures than at cooler temperatures. We saw this in our borax experiment. We could only dissolve less than a gram of borax in 100 ml of water at ice cold temperatures (about 2 degrees C) before the solution became saturated and the borax I added would no longer dissolve. But if we heated the water to near boiling (almost 80-90 degrees C), we could dissolve almost 59 grams of borax before the solution was saturated. Why is this?
If you remember when we discussed density last year, we noticed that adding thermal energy causes particles to vibrate more and become less dense (or more loosely packed). Heating the water in our experiment lowered the density of the water and created more space between the solvent (water) molecules to allow the solute (borax) to dissolve in between the spaces. But as the solvent cooled, it became more dense (more tightly packed), which caused some of borax molecules to get "squeezed out" of (or precipitate from) solution, forming crystals. This change in density causes the concentration at which the solution is saturated to decrease with decreasing temperature.
This experiment demonstrated 2 things:
In general, you can dissolve more solute at higher temperatures than at cooler temperatures. We saw this in our borax experiment. We could only dissolve less than a gram of borax in 100 ml of water at ice cold temperatures (about 2 degrees C) before the solution became saturated and the borax I added would no longer dissolve. But if we heated the water to near boiling (almost 80-90 degrees C), we could dissolve almost 59 grams of borax before the solution was saturated. Why is this?
If you remember when we discussed density last year, we noticed that adding thermal energy causes particles to vibrate more and become less dense (or more loosely packed). Heating the water in our experiment lowered the density of the water and created more space between the solvent (water) molecules to allow the solute (borax) to dissolve in between the spaces. But as the solvent cooled, it became more dense (more tightly packed), which caused some of borax molecules to get "squeezed out" of (or precipitate from) solution, forming crystals. This change in density causes the concentration at which the solution is saturated to decrease with decreasing temperature.
This experiment demonstrated 2 things:
- How crystals form when a solution cools and exceeds the new, lower saturation concentration. Remember that there is still some borax dissolved at the lower temperature, only less of it because the saturation concentration is now lower.
- We ALSO noted how crystal size is affected by the speed of cooling. If we cooled the hot solution quickly in a ice bath, the crystals were smaller than those in the solution we cooled more slowly at room temperature. By quickly cooling the solution, the solute precipitates rapidly, which gives the particles less time to travel to and build on the crystal lattice.
Using a sponge to MODEL Saturation & Precipitation from Solution by Cooling
During class, I used a sponge to model saturation AND precipitation from solution by cooling. In this case, the sponge models the solvent and water models the solute. (Normally water is the solvent!)
- First, I added water to the sponge until the sponge could absorb no more water. This demonstrated that the sponge was fully saturated. Image #2 best illustrates this
- If I added any more water, it would simply drip (or 'precipitate') out of the sponge. Image #5 best illustrates this
- When I squeezed the sponge, more water poured out of the sponge. I was increasing the density of the sponge using pressure! Remember from last year's studies that density can be changed by changing the temperature (thermal energy) or changing the pressure. In this case, I was using pressure, but you can use this as a model for precipitation from solution by cooling, because I can also increase the density by reducing the amount of thermal energy (lowering the temperature) of the solvent. Images #7 & 8 best illustrate this
Click to set custom HTML
How to make sugar crystals (cooling a saturated solution)
|
The video on the right shows how to saturate a solution with sugar and then let it cool. Because you can dissolve more particles into a heated solution than in a cool solution, the process of cooling "kicks out" or precipitates the sugar molecules by lowering the saturation concentration, which form a precipitate in the form of sugar crystals.
Note: If you choose to make rock candy, make sure a parent supervises. Hot sugar solution can burn! Also, I recommend that you cool the solution SLOWLY by wrapping it in a towel...WHY??? |
|
Slower cooling or evaporation results in larger crystals
Recrystallization: The 3rd method of forming crystals
|
3. Recrystallization: High temperature and/or pressure changes (or metamorphoses) an existing mineral and results in 2 possible outcomes
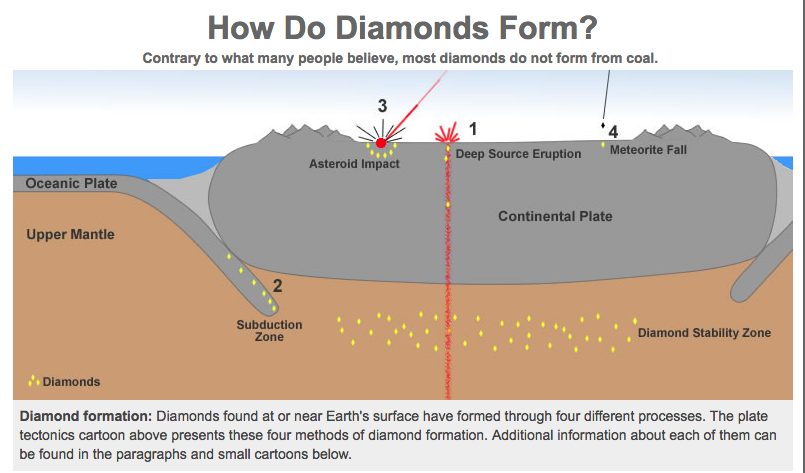
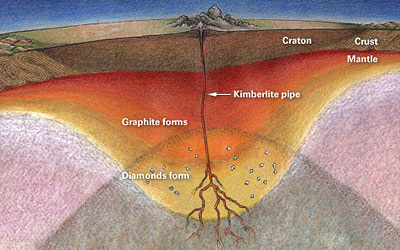
a. Ex. when snow compacts & becomes ice (same mineral, different appearance or 'habit') b. Diamonds, which form under extremely high pressures in the mantle, can recrystallize into graphite when the pressure decreases if the diamond rises to the earth's surface slowly instead of quickly Sooo...even though some diamonds were formed 3 billion years ago, they AREN'T forever! c. Ex. Calcite and aragonite can recrystallize into each other (Even though they are composed of the same particle, the conditions cause the crystal system to change so they become different minerals - see below) Note: This is an exception to the rule that crystals form from a fluid (meaning - either the liquid form of the particle, or a particle in solution) because recrystallization occurs without fluid - or in the solid state. In other words, it ALREADY starts out as a solid crystal. Here, the particles are subjected to such extreme pressure and/or temperature that they change (or metamorph) into EITHER :
This means the same mineral can simply be compacted (snow changes "habits" into ice), OR that one mineral changes into an entirely different mineral by changing its crystal system (see D. below and the example of how calcite and aragonite can recrystallize into each other). |
Coal does NOT make Diamonds
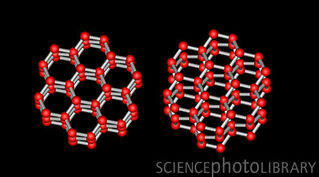
It's a common misconception that coal is the source of diamonds, but the evidence suggests that is not the case. If you are interested, you can go to this Geology.com webpage, which discusses the evidence. While it is theoretically possible to make diamonds out of any carbon-rich source with enough pressure (even peanut butter!), the cost required to do so would cost more than the resulting diamond itself. Graphite can be formed from coal. Diamonds actually form by solidification in the mantle. Because they are under extreme temperature and pressure, diamonds form in the cubic system, but not by recrystallization of coal or graphite.
|
Recrystallization of Calcite and Aragonite
|
Aragonite and calcite are typically the components of shells and corals. They are considered polymorphs (poly=many, morph = form or shape) of each other, even though they are different minerals. This is because they share the same composition (CaCO3), but different crystal systems (see below). They're a great example of how one mineral can recrystallize into another by changing crystal systems without melting or going into solution. No fluids involved!
With extreme pressure, calcite will recrystallize into aragonite. But, because aragonite is relatively unstable, it will slowly recrystallize into calcite in seawater - without ever becoming a fluid. You can also cause aragonite to recrystallize into calcite at high temperatures (400o F) under dry conditions. |
FOR THE NOTES UNDER "D", you should be able to tell me (for quiz purposes)
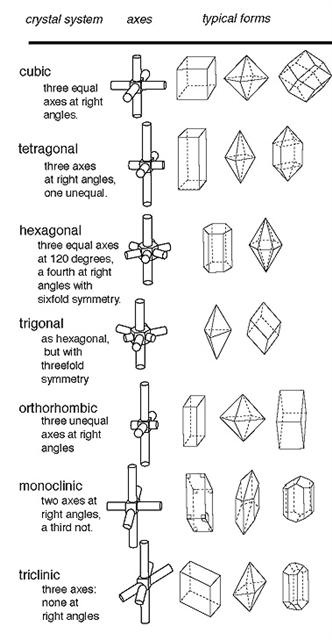
D. The shape or structure of the lattice pattern is called a crystal system
- what an axis of symmetry is and
- what is the same and what is different when comparing the cubic and tetragonal crystal systems.
D. The shape or structure of the lattice pattern is called a crystal system
|
1. Think of them as building blocks
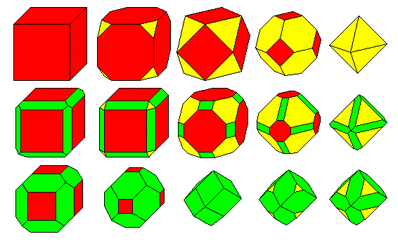
2. There are 6 (or 7) basic crystals systems that are identified by the symmetry and length of their axes (axes of symmetry): (Click on the above note to go to a link that explains an axis of symmetry) a. Cubic: all 3 axes are the same length & perpendicular b. Tetragonal: 2 axes are equal length while a third axis is a different length, all axes are perpendicular to each other c. Hexagonal and Trigonal: 3 of the 4 axes are the same length & perpendicular to the 4th axis, which is a different length i. Some scientists classify these as 2 separate systems, because the hexagonal system is 6-sided and the trigonal system is 3-sided; but they both share the same axes of symmetry, which is why they are often classified in the same crystal system d. Orthorhombic: all 3 axes are different lengths and are perpendicular to each other e. Monoclinic: Each of 3 axes is a different length; 2 are at right angles to each other and the third is inclined f. Triclinic: All 3 axes are of different lengths and inclined towards each other |
Crystal Forms
How the crystal grows depends on the conditions under which it was formed............
|
4. Which crystal system and which habit the crystal will adopt depends on a variety of conditions during its formation, including:
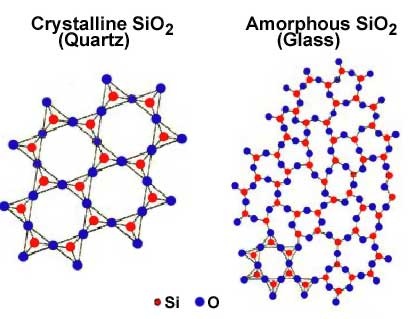
a. the temperature and how quickly or slowly the fluid cools or evaporates i. Generally, the slower the cooling or evaporation, the larger the crystal. ii. For example, if SiO2 (quartz) cools slowly, it will form rock crystal; but if it cools more rapidly, it will form agate. iii. Fluids that cool so rapidly that the particles don't have time to travel to their place on the lattice will not form a crystal and are called amorphous (a = without, morph = form) and are also known as a glass They are NOT called crystals because the particles do not form a regular, repeating pattern before they solidify. We call them amorphous (without form) rocks. Window glass is an example of quartz sand that was melted and cooled rapidly. Another example is obsidian, which results from lava cooling rapidly. Frothy lava may cool into a glass known as pumice. |
|
|
. b. the pressure under which it forms
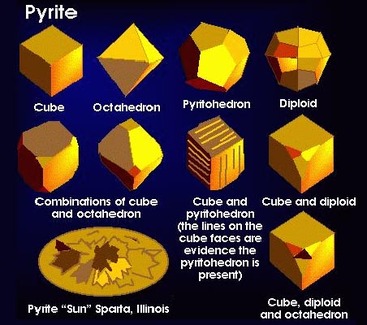
i. Diamond and graphite are made of the same element, carbon, but form their crystal lattice under differing pressures Explanation (not part of notes) Carbon crystals formed under extremely high pressure deep in the Earth’s mantle form cubic lattices that make diamonds. Graphite forms hexagonal crystal lattices from the metamorphism of carbon sediments. c. the type of particle the crystal is made of i. A single particle can form into some, but not all crystal systems ii. Ex. FeS2 can form cubic (pyrite) or orthorhombic (marcasite) crystal lattices , but it cannot form into other crystal lattices (e.g., monoclinic or hexagonal) See examples of pyrite and marcasite on the right. Note: when you read about polymorphism below, you will see that pyrite and marcasite are polymorphs of each other. |
|
Polymorphism
|
5. Depending on the conditions under which the crystal is formed, the same particle can crystallize in different crystal systems and the same crystal can have different habits
a. Called polymorphism (poly = many; morph = form) |
NOTE! Polymorphism can arise from two different circumstances:
1) When the same particle crystallizes in different crystal systems and result in two different minerals; OR
2) When the same crystal (same particle, same crystal system) forms different habits
Even though polymorphism can arise in all 3 methods of crystallization, if you think about it, recrystallization always results in polymorphs.
1) When the same particle crystallizes in different crystal systems and result in two different minerals; OR
2) When the same crystal (same particle, same crystal system) forms different habits
Even though polymorphism can arise in all 3 methods of crystallization, if you think about it, recrystallization always results in polymorphs.
E. Crystals are the microscopic building blocks of minerals.