|
|
|
|
b. Chemical
i. Precipitation of dissolved sediments from solution by evaporation ii. Ex. travertine limestone, rock salt, stalagtites Limestone comes in many forms, but it is composed of at least 50% calcium carbonate in the form of calcite or aragonite. One type of limestone called travertine limestone is produced when water that travels underground through limestone rock dissolves the limestone and, as the water evaporates, the limestone is reformed (much like your halite experiment and the dissolved salt precipitated from solution by evaporation).
In the case of the Mammoth Hot Springs (above right), the springs emerge and the hot water evaporates, the limestone precipitates and deposits on the Mammoth springs terraces. The stalactites and stalagmites in the Carlsbad Caverns (above) are also travertine limestone. AND, the lime deposits in your toilet are the same thing!!! |
|
|
c. Organic
i. Formed primarily from remains of once-living organisms ii. Ex. coal (decayed vegetation); chalk & fossiliferous limestone (skeletons & shells of marine organisms) |
Can limestone be both organically or chemically formed sedimentary rock?
YES!! Limestone comes in many forms and can be organic or chemical sedimentary rock. Either way, it is composed of at least 50% calcium carbonate (CaC03) in the form of calcite (trigonal) or aragonite (orthorhombic). So what's the difference between organically formed limestone and chemically formed limestone? Organic limestone results from the compaction and cementation of accumulated piles of marine shells, bones, coral, algal diatoms, foraminifer tests (kind of a shell) and even poop! Chemical limestone results from the evaporation of water that contains dissolved limestone (travertine), or the precipitation (falling out of solution) of calcite or aragonite from waters that are supersaturated with dissolved limestone (tufa). Click here for a full explanation and some other examples of the different types of limestone.
Organic limestone
|
Chemical limestone
|
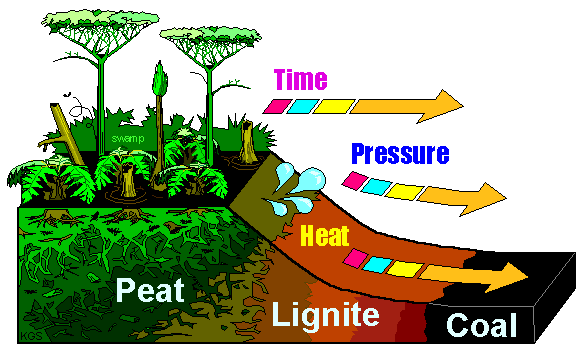
How coal is formed...and metamorphosed?
|
Here's some interesting facts about coal. It's called a 'fossil fuel' because we burn coal to produce energy and because it comes from "fossil plants". So how does coal form from prehistoric plants?
Hundreds of millions of years ago (during the Carboniferous Period) the Earth had large areas covered with swampy bogs where giant dragonflies flew and prehistoric trees and plants grew. In the eastern U.S., the bogs were sea water bogs that contained sulfur. After they died, and the sea dried up, layers of sand and rock covered and compressed the dead plants in sedimentary layers along with the sulfur. In the western U.S., the ancient bogs were freshwater, and contained less sulfur. With relatively low pressure from the overlying sediments, the dead plant material compresses into peat. The water in the bogs slows down the plant decaying process, so the dead plants are buried faster than they decay. Peat is low density and can be dried out and burned. As more layers form above, the peat is compacted to lignite, or brown coal, and with yet more layers (and pressure) becomes a more dense bituminous coal. At this point it is still considered a sedimentary rock. But with increased heat and pressure, the bituminous coal becomes anthracite (see progression on right), which is a very dense form of coal that is now considered a metamorphic rock. At this point, it is almost pure carbon. Now, add even more heat and pressure to make it so dense that it does not burn well. But it does become easy to write with - because it is now graphite! (Remember that metamorphosis can result in recrystallization!) |
A slide show at this link explains the steps in coal formation over time.
You may think that additional heat and pressure would turn the graphite (or the carbon rich rock, coal) into diamonds, but this is not what happens. Click here to find out why diamonds do not naturally form from coal!
|
How much coal do you use? (a little Earth Day note)
Currently, coal is mined in 26 of the 50 states and nearly half of the electricity you use comes from power plants that use coal. {Most of Madison's power comes from the Columbia power plant, which burns low sulfur coal which is shipped from out west by train - which, of course, uses fuel, too.} Because of this, the average family uses literally tons of coal each year. Consider this....an electric water heater alone gets it energy from burning about 2 tons of coal per year. Your refrigerator uses 1/2 ton/year, and your stove uses another 1/2 ton!
Burning coal puts additional carbon dioxide into the air, which contributes to global warming and ocean acidification. Burning high sulfur coal (from those sea swamps mentioned earlier), adds sulfur dioxide into the air, which leads to acid rain. Remember how we talked about how you can dissolve limestone with acid, and how limestone is made from the sedimentary deposits of shells? If you are a live organism with a shell (coral, diatoms, shell fish, etc.) living in an acidified ocean, your shell dissolves, too! This is why coral reefs, which are some of the most biodiverse regions in the world, are threatened with extinction.
Reducing your electricity consumption lowers the amount of coal you burn!
Burning coal puts additional carbon dioxide into the air, which contributes to global warming and ocean acidification. Burning high sulfur coal (from those sea swamps mentioned earlier), adds sulfur dioxide into the air, which leads to acid rain. Remember how we talked about how you can dissolve limestone with acid, and how limestone is made from the sedimentary deposits of shells? If you are a live organism with a shell (coral, diatoms, shell fish, etc.) living in an acidified ocean, your shell dissolves, too! This is why coral reefs, which are some of the most biodiverse regions in the world, are threatened with extinction.
Reducing your electricity consumption lowers the amount of coal you burn!